COVID-19 Vaccine Eye Injuries: Ocular Adverse Events, Vision Problems, Blurry Vision, Visual Impairment, Blindness and More

As of March 9, 2023, WHO’s VigiAccess database recorded 158,649 Adverse Events involving “Eye disorders” following COVID-19 vaccination: (click here)

Most notable are:
- Vision blurred: 32,935
- Visual impairment: 25,413
- Blindness 3,982
- Blindness, unilateral 1,370
In 2021, a total of 717,577 COVID-19 vaccine adverse events were reported to VAERS (click here), and 55,313 of those involved ocular side effects.
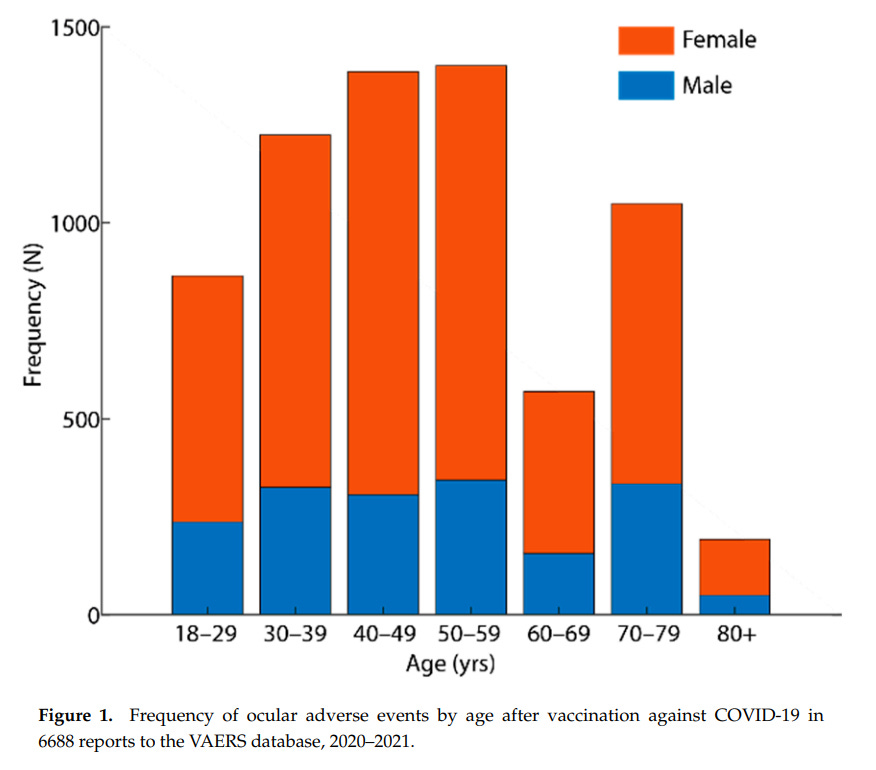
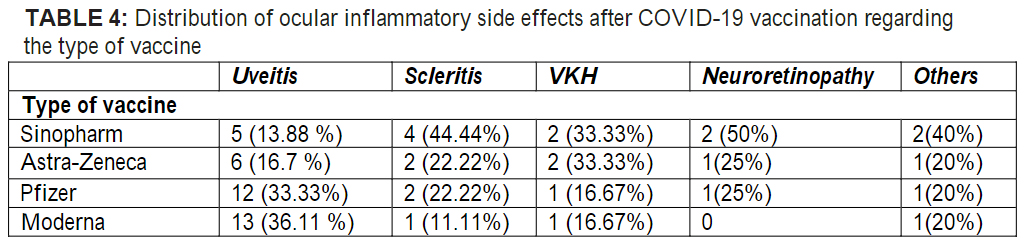
One study examined 6688 of these VAERS ocular side effects (Nyankerh et al.)
- 74% of adverse event reports were women
- age group 40 to 59 years old had most frequent adverse events
- age group 18 to 29 had a fairly very high frequency of adverse events
- Moderna and J&J vaccines had higher proportion of adverse ocular events
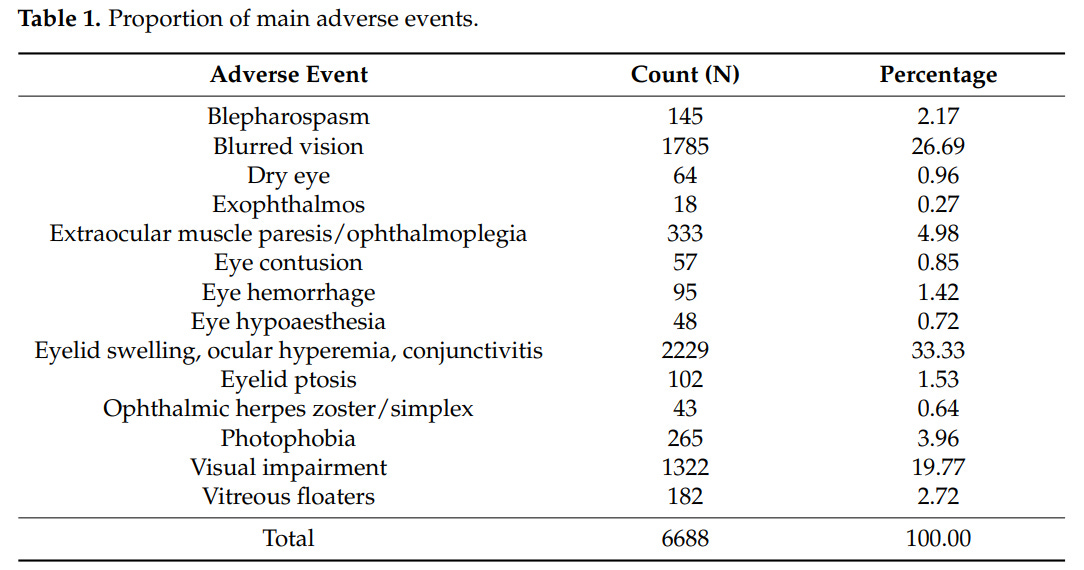
Top 3 ocular adverse events out of the 6688 cases examined:
- eyelid swelling, ocular hyperemia, conjunctivitis (33.3%) – 2229 cases
- blurred vision (26.7%) – 1785 cases
- Visual impairment (19.8%) – 1322 cases
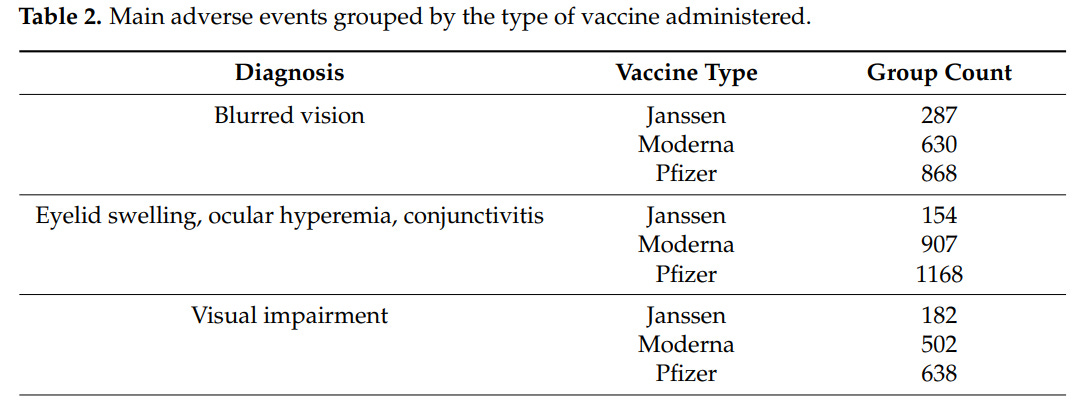
A Spanish study looked at 70 cases of ocular side effects post COVID-19 vaccine (Pfizer, Moderna, AstraZeneca) (click here)
- 57% were women
- average age was 51
- 50/70 (71%) had uveitis, 10% scleritis, 19% other
- Most concerning were 5 out of 65 patients (7.2%) who had reduced visual acuity
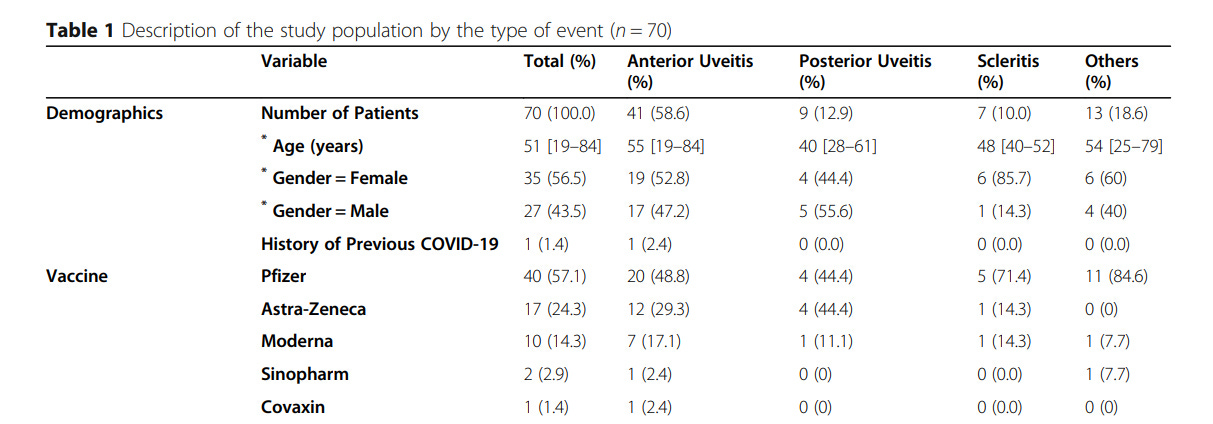
A study from Iraq, published Feb.2023, examined 60 cases with ocular side effects post COVID-19 vaccine (Pfizer, Moderna, AstraZeneca). (click here).
- 53% were women
- 36/60 (60%) had uveitis, 15% scleritis, 10% VKH, 7% Neuroretinopathy, 8% other
- Most concerning were 4 out of 60 patients (6.7%) who had loss of visual acuity

Government of Canada lists more types of ocular adverse events and eye injuries following COVID-19 vaccination, than for COVID-19 infection (click here):

- Autoimmune reactions (eg. uveitis, optic neuritis, Graves) (click here)(click here)
- Viral Reactivation of herpes, varicella zoster virus (click here)
- Parasite reactivation (toxoplasma) (click here)(click here)
- Blood clots (eg. retinal artery/vein occlusion) (click here)(click here)
- Blood vessel inflammation (eg. retinal vasculitis, Behcet’s) (click here)(here)
- Bleeding or hemorrhage (eg. retinal hemorrhage) (click here)
- Demyelination (eg. multiple sclerosis lesions in the brain (click here)(click here)
- Reaction to Polyethylene glycol (PEG)-2000 lipid in mRNA vaccines (click here)
- Corneal graft rejection (click here)
Epoch Times ran an excellent article on Dec.16, 2022 called: “Eye Problems? You may have been blinded by your trust of COVID-19 vaccines” (click here):
Dr. Lynnell Lowry is an ophthalmologist in private practice in San Antonio, Texas. She said that she has seen more unusual eye problems since the roll-out of the COVID-19 vaccines than she ever has in 25 years of practicing medicine.
“A lot of folks are coming in with a visual disturbance that we can’t explain”
COVID-19 vaccines have adversely affected the eyes of 100,000s of people and 10,000s have suffered permanent visual impairment up to and including blindness.
These are often very serious side effects and unfortunately, they are common.
Women suffer eye injuries more often than men, and age group 40-59 is most impacted. Autoimmune reaction seems to be the most common mechanism of COVID-19 vaccine injury.
As with all other adverse events following COVID-19 vaccination, the side effects involving eyes have been downplayed by health authorities and doctors.
A thorough discussion of the risks of eye injury should be conducted as part of informed consent, with any doctor who recommends COVID-19 vaccination.
*
Note to readers: Please click the share buttons above. Follow us on Instagram and Twitter and subscribe to our Telegram Channel. Feel free to repost and share widely Global Research articles.
Dr. William Makis is a Canadian physician with expertise in Radiology, Oncology and Immunology. Governor General’s Medal, University of Toronto Scholar. Author of 100+ peer-reviewed medical publications.
All images are from the author